ITOCHU Announces that A2 Healthcare Corporation Establishes Boston Branch in the United States, Supporting Biotech Companies in the Development of New Drugs for Japan
March 19, 2024
ITOCHU Corporation (headquartered in Minato-ku, Tokyo; Keita Ishii, President & COO; hereinafter “ITOCHU”) announced today that the subsidiary A2 Healthcare Corporation (headquartered in Bunkyo-ku, Tokyo; Hitoshi Kamiya, President & CEO; hereinafter “A2 Healthcare”) has become Japan’s first major CRO*1 to establish a Boston Branch in the United States, doing so on March 1, 2024 to expand its business in the US, commencing support of biotech companies in the development of new drugs in Japan.
Currently, overseas pharmaceutical companies shy away from the Japanese market due to Japan’s strict pharmaceutical regulations, complex clinical trial-related procedures, and drug pricing system that differs from Europe and the United States, leading to the expansion of “drug loss” since fewer new drugs enter the Japanese market. The proportion of “domestically unapproved drugs”, which drugs that have been approved in Europe and the United States but not in Japan is on the rise (56% in 2016, 72% in 2020).*2 Moreover, more than half of the domestically unapproved drugs are developed by overseas biotech companies*3. Thus, in order to eliminate drug loss, there is a need for a new mechanism to motivate overseas companies to develop drugs in Japan.
A2 Healthcare has been entrusted with a large number of sponsor-initiated clinical trials and investigator-initiated clinical study in various fields and diseases, possessing top-class know-how in the industry to support new drug development. In October last year, A2 Healthcare received an award in the “Best CRO/CDMO” category of the Citeline Pharma Intelligence Awards Japan 2023,*4 as been recognized for its know-how in efficient recruitment of subjects for clinical trials in Japan and the advanced use of IT in clinical trials.
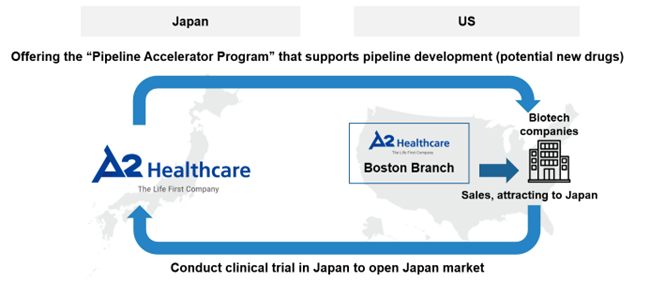
A2 Healthcare has now established a branch in the Boston metropolitan area, which is part of the world's largest biotech cluster with more than 1,000 biotech companies, to secure contacts with overseas companies and support the development of new drugs in Japan. Specifically, it offers the Pipeline Accelerator Program, a financing program that provides financial support for the development of companies’ pipelines (potential new drugs). Moreover, it consistently provides CRO services for new drug development in Japan and supports the development of new drugs that are in high demand among Japanese patients.
ITOCHU transforms business through its "market-in" approach, which is a basic policy included its medium-term management plan, and this effort is in line with this policy. ITOCHU and A2 Healthcare will put our efforts into solving the social issues of drug lag and drug loss and contribute to society by improving healthcare through this initiative focused on the healthcare field.
- *1CRO: An abbreviation for "Contract Research Organization" and an outsourced business engaged in work required in the development of pharmaceutical products (monitoring, data assessment, statistical analysis, etc.)
- *2Source: Japan CRO Association, "Japan CRO Association Annual Report 2023"
- *3Source: “Drug Lag: Situation and Characteristics of Unapproved Drugs in Japan,” Office of Pharmaceutical Industry Research (OPIR)
- *4Japanese version of Citeline's Scrip Awards, the most prestigious awards in the pharmaceutical industry. They assess service quality and the building of relations with client companies.
|
|
Outline of the A2 Healthcare US Boston Branch (A2US)
| Name (provisional name) | A2 Healthcare Corporation US Boston Branch |
|---|---|
| Location | One Broadway 14th Floor, Cambridge, MA 02142 |
| Branch general manager | Hiroki Matsushima |
A2 Healthcare Corporation
| Location | 20F, Sumitomo Korakuen Bldg., 1-4-1 Koishikawa, Bunkyo-ku, Tokyo |
|---|---|
| Representative | Hitoshi Kamiya |
| Established | July 2003 |
| Business | Clinical development, post-marketing surveillance and clinical research services for development of pharmaceutical, medical devices, cellular and tissue-based products and vaccines |
| URL | https://www.a2healthcare.com/ en/ |